Pretreatment Human Immunodeficiency Virus (HIV) Drug Resistance Among Treatment-Naive Infants Newly Diagnosed With HIV in 2016 in Namibia: Results of a Nationally Representative Study
Michael R Jordan, Leonard Bikinesi, Laimi Ashipala, Nicholus Mutenda, Mary Brantuo, Gillian Hunt, Andreas Shiningavamwe, Gram Mutandi, Anita Beukes, Suzanne Beard, Katherine Battey, Eric J Dziuban, Elliot Raizes, Paul Adjei, Alice Tang, Amalia Giron, Steven Y Hong
Open Forum Infectious Diseases, Volume 9, Issue 5, May 2022, ofac102,
PMID: 35434174 | PMCID: PMC9007920 | DOI: 10.1093/ofid/ofac102
Abstract
Background: The World Health Organization (WHO) recommends routine surveillance of pretreatment human immunodeficiency virus (HIV) drug resistance (HIVDR) in children <18 months of age diagnosed with HIV through early infant diagnosis (EID). In 2016, 262 children <18 months of age were diagnosed with HIV in Namibia through EID. Levels of HIVDR in this population are unknown.
Methods: In 2016, Namibia surveyed pretreatment HIVDR among children aged <18 months following WHO guidance. Reverse transcriptase, protease, and integrase regions of HIV-1 were genotyped from remnant dried blood spot specimens from all infants diagnosed with HIV in Namibia in 2016. HIVDR was predicted using the Stanford HIVdb algorithm.
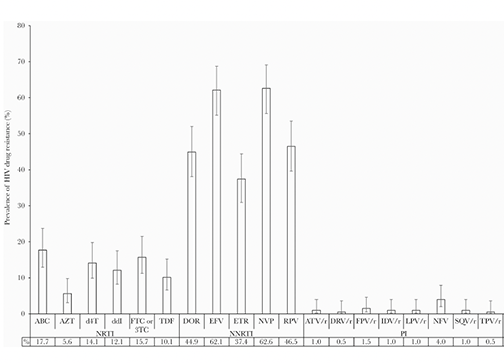
Results: Of 262 specimens genotyped, 198 HIV-1 protease and reverse transcriptase sequences and 118 HIV-1 integrase sequences were successfully amplified and analyzed. The prevalence of efavirenz/nevirapine (EFV/NVP), abacavir (ABC), zidovudine, lamivudine/emtricitabine (3TC/FTC), and tenofovir (TDF) resistance was 62.6%, 17.7%, 5.6%, 15.7%, and 10.1%, respectively. No integrase inhibitor resistance was detected.
Conclusions: The high level of EFV/NVP resistance is unsurprising; however, levels of ABC and TDF resistance are among the highest observed to date in infants in sub-Saharan Africa. The absence of resistance to dolutegravir (DTG) is reassuring but underscores the need to further study the impact of ABC and 3TC/FTC resistance on pediatric protease inhibitor- and DTG-based regimens and accelerate access to other antiretroviral drugs. Results underscore the need for antiretroviral therapy optimization and prompt management of high viral loads in infants and pregnant and breastfeeding women.
Keywords: HIV; Namibia; drug resistance; infants.